Graduate student Vijay Jupally will present “Synthesis, Characterization and Electrochemical Properties of Gold Nanoparticles” to the department.
Abstract:
Ultra-small gold nanomolecules are <3 nm in size and comprise a precise number of gold atoms and thiolate ligands.These novel materials find applications in the fields of drug delivery, 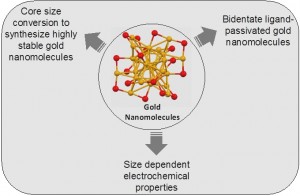 sensing, catalysis and photovoltaics. In this talk three topics will be covered: (1) Interstaple dithiol cross-linking in Au25(SR)18 nanomolecules. (2) Core size conversion in gold nanoclusters and (3) Electrochemical properties of gold nanomolecules. First, bidentate dithiolate ligands were incorporated on the surface of Au25(SR)18 nanomolecules via ligand exchange reactions. The affinity and binding of various dithiols was systematically studied by varying the carbon chain length between the thiol groups and the affect on the properties was studied.1 Second, core size conversion of larger and smaller gold nanomolecules to synthesize highly stable nanomolecules was studied. Highly stable Au130(SR)50 was synthesized with several ligands and alloys via core size conversion of nanoclusters >40 kDa.2 Similar core size conversion reactions in nanoclusters <30 kDa were also investigated.3 Third, the size dependent electrochemical properties of gold nanomolecules were studied. The variation of electrochemical gap, representative of the HOMO-LUMO gap, was studied using cyclic voltammetry and differential pulse voltammetry for different sizes. Electrochemical results showed the molecule-like to bulk metal-like transition in gold nanomolecules.4
sensing, catalysis and photovoltaics. In this talk three topics will be covered: (1) Interstaple dithiol cross-linking in Au25(SR)18 nanomolecules. (2) Core size conversion in gold nanoclusters and (3) Electrochemical properties of gold nanomolecules. First, bidentate dithiolate ligands were incorporated on the surface of Au25(SR)18 nanomolecules via ligand exchange reactions. The affinity and binding of various dithiols was systematically studied by varying the carbon chain length between the thiol groups and the affect on the properties was studied.1 Second, core size conversion of larger and smaller gold nanomolecules to synthesize highly stable nanomolecules was studied. Highly stable Au130(SR)50 was synthesized with several ligands and alloys via core size conversion of nanoclusters >40 kDa.2 Similar core size conversion reactions in nanoclusters <30 kDa were also investigated.3 Third, the size dependent electrochemical properties of gold nanomolecules were studied. The variation of electrochemical gap, representative of the HOMO-LUMO gap, was studied using cyclic voltammetry and differential pulse voltammetry for different sizes. Electrochemical results showed the molecule-like to bulk metal-like transition in gold nanomolecules.4
References:
(1) Jupally, V. R.; Kota, R.; Dornshuld, E. V.; Mattern, D. L.; Tschumper, G. S.; Jiang, D.-e.; Dass, A. J. Am. Chem. Soc. 2011, 133, 20258.
(2) Jupally, V. R.; Dass, A. Phys. Chem. Chem. Phys. 2014, 16, 10473.
(3) Nimmala, P. R.; Jupally, V. R.; Dass, A. Langmuir 2014, 30, 2490.
(4) Jupally, V. R.; Thrasher, J. G.; Dass, A. Analyst 2014, 139, 1826.